- Services
- /
- Covid-19
Covid-19
Services & Care
You can count on Sollis to provide a full spectrum of care including prevention, testing and cutting-edge treatment for Covid-19 and Long Covid.
Covid-19 Services

Testing & Vaccines
Members receive unlimited rapid & PCR testing. We also provide vaccinations including boosters.
Travel Coordination
We navigate travel regulations, testing requirements and provide all necessary documentation.
Event Services
With on-site rapid event testing, you can host and gather safely at your home or a private venue.
At-Home Care
Monoclonal antibody treatment, oxygen therapy, and remote monitoring to ensure swift recovery.
RITA W.
Sollis Member
During Christmas my entire family had Covid, including my mother who is 92. Your team came to the house and treated us all – I really can’t thank you enough. Sollis has truly been a godsend to all of us.”
Interested in Sollis?
EVENT SERVICES
From intimate gatherings to large-scale conferences, our on-site testing and event services provide an additional level of safety and peace of mind to both you and your guests.

JEN ARMSTRONG
COO of Baby2Baby
The services Sollis Health provided for the Baby2Baby 10 year Gala helped to ensure a safe and successful night.”
Membership Plans
In order to ensure the highest standard of care,
we maintain a limited number of memberships.


MEMBERSHIPS
START AT
Over 45 — $5,000
Under 45 — $3,000
18 & Under — $1,000
*Ask about tiered family pricing.
Interested in Sollis Membership?
Sollis is a members-only medical concierge service.
Discover a Sollis
Center near you.
Upper East Side Flagship
170 E 77th St, Lower Level
Open 24/7, 365.

Fifth Avenue Center
945 Fifth Avenue
Mon-Sun: 7am-7pm

Tribeca Center
255 Greenwich St, 5th Floor
Mon-Sun: 7:30am-7:30pm
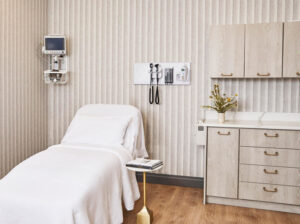
Gramercy Center
11 East 22nd Street, 3rd Floor
Mon-Fri: 9:00am-5:00pm
Sat & Sun: Closed

Water Mill Center
760 Montauk Highway,
Suite 2C
Mon-Sun: 9am-5pm

Beverly Hills Flagship
155 N San Vicente Blvd
Open 24/7, 365

Santa Monica Center
1243 7th St, Suite B
Mon-Fri: 7am-11pm
Sat & Sun: 8am-8pm

Manhattan Beach Center
1000 N. Sepulveda Blvd
Mon-Fri: 7am-7pm
Sat & Sun: 8am-5pm
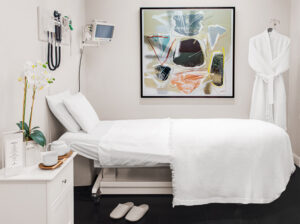
San Francisco Flagship
1005 Van Ness Ave
Open 24/7, 365

Palm Beach Flagship
324 Royal Palm Way,
Suite 100
Open 24/7, 365.

Boca Raton Center
1905 Clint Moore Rd #303
Mon-Sun: 8am-5pm
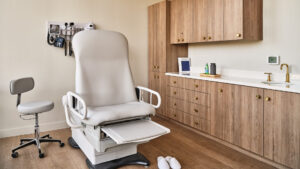
Miami House Call Program
Miami-Dade and Broward County Area
(305) 824-2300





